What Are the Best Nootropics? The Top 7 Powerful Nootropics

“Smart drugs,” often referred to as nootropics, are synthetic compounds or natural substances (i.e. plant extracts) that have been shown in studies to improve overall mental performance and well-being.
Nootropics have become a hot topic, and are in high demand, due to an increasingly competitive society wherein many people choose to research and experiment with these compounds, often referred to as “brain hacking” or “biohacking.” The primary nootropic effects that people are interested in are improved memory, improved creativity, improved concentration, motivation, and focus, better sleep, and an overall increase in well-being and positive mood. Based on reviews and reports, many people seem to be on a quest to find the best nootropics stack (combination), if they choose to consume these substances.
In this nootropic review, we’ll discuss some of the most popular and powerful nootropic compounds, how they work, and a summary of available research and anecdotal evidence to support the nootropic benefits as well as side effects.
- Caffeine
- L-Theanine
- Phenibut HCL
- Tianeptine Sodium
- DMAA (1,3- dimethylamylamine )
- DMHA (Octodrine)
- Nicotine
Disclaimer: ELV Bioscience (www.elvbio.com) does not make any recommendations for use, inclusive of the following compounds in this review of popular and potent compounds with nootropic effects.
Caffeine:
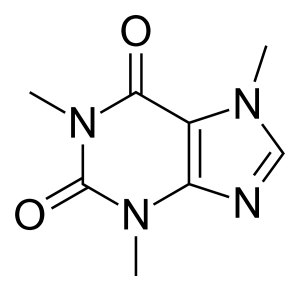
Caffeine is the most widely used psychoactive substance in the world. Caffeine is an alkaloid most often consumed in the form of coffee, tea, guarana, chocolate, and kola nuts. However, caffeine is also consumed in supplemental form, whether it is extracted and isolated from a natural source, such as pure caffeine isolate (from green tea) by ELV Bioscience, or whether it is made synthetically. Many popular supplemental sources of caffeine include pre-workout products, energy drinks, and energy shots. Caffeine is a central nervous system stimulant in the methylxanthine class of compounds.
How Caffeine Works?
Caffeine is a central nervous system stimulant, which works by way of blocking (antagonizing) adenosine receptors in the brain, causing a sense of alertness. Adenosine is a naturally occurring molecule that accumulates throughout the day to help slow down neurotransmission, promote a sense of calm and tiredness, and aid in supporting the natural circadian rhythm.
Caffeine Benefits:
Research has shown that caffeine consumption, within a recommended dose range, can have several benefits. Studies have shown that the benefits of caffeine can include possible protection against Alzheimer’s disease and dementia, improved exercise output and performance, and improved erectile function in men. Based on available research
Caffeine Side Effects:
Common side effects of excess caffeine consumption can include feeling jittery or anxious as well as difficulty sleeping due to caffeine’s long half-life of 5-6 hours. Other side effects of excessive caffeine consumption can include elevated heart rate and blood pressure due to vasoconstriction. Common side effects of caffeine withdrawal from prolonged consumption can include fatigue, malaise, and headache.
Caffeine Dosage:
Several health authorities, including the Mayo Clinic, Harvard, and the American Heart Association, suggest that consuming up to 400mg of caffeine per day in healthy adults is considered safe.
L-Theanine:
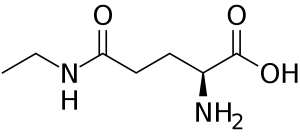
L-Theanine is a non-essential amino acid that naturally occurs in the select tea plants. The most popular natural source of theanine is Camellia Sinensis, which is a plant from which white, green, and black teas are made. Guayusa is another plant that contains theanine. Theanine is also consumed in supplement form as well, whether it is naturally isolated from a tea plant, or whether it is synthetically produced. Theanine is one of the best nootropics, which has become very popular in recent years. This Theanine review will focus on L-Theanine, which is the naturally occurring enantiomer. D-Theanine is the opposite enantiomer but does not have as much research supporting its safety and efficacy.
How Theanine Works?
L-Theanine natural nootropic that is a structural analogue of two other naturally occurring amino acids, L-Glutamine, and L-Glutamate. Theanine is the primary contributor to “umami” (savory) flavor notes found in green tea.
Theanine is water-soluble, absorbs through the small intestine, and passes through the blood-brain barrier intact. One in the brain (central nervous system) Theanine binds to glutamate receptors since as we discussed earlier, it is structurally similar to the amino acid neurotransmitter – glutamate. Glutamate is an excitatory (stimulating) neurotransmitter. Since Theanine binds to glutamate receptors, specifically AMPA and kainate receptors, and the NMDA receptor, it can block some of the excitatory effects of glutamate. Theanine also blocks the reuptake of glutamine and glutamate by blocking their respective transporters. This is why many people who consume Theanine, whether in smaller amounts from tea or in larger amounts from supplements, often report that it promotes a sense of calm.
Theanine has been shown to increase levels of dopamine, GABA, glycine and serotonin levels in certain areas of the brain. Currently, the available research is not fully clear on Theanine’s effect on serotonin given that some research shows an increase and other research shows a decrease in serotonin concentrations. Theanine has also been shown to increase levels of brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF) in various studies. L-Theanine has a half-life of approximately 3-4 hours following oral administration.
Theanine Benefits:
Theanine is often reported to promote a sense of calm, anxiety/stress relief, improved mood, improved focus, and the ability to create synergy with caffeine by reducing caffeine side effects. Research shows that Theanine may be able to inhibit glutamate excitotoxicity. Theanine also possesses antioxidant properties, which can promote both the central nervous system and systemic benefits.
Theanine Side Effects:
While Theanine is considered safe by many regulatory authorities including the US FDA, some people do report side effects. Some common side effects may include fatigue, “brain fog,” or in more rare cases, a paradoxical effect wherein Theanine can promote an anxious state.
Theanine Dosage:
A research organization named Natural Standard has suggested that Theanine is safe up to 1,200mg per day. The most common supplemental dosages are 100mg-200mg. Theanine content in tea can range from approximately 8mg to approximately 50mg in shade-grown teas such as Matcha green tea.
Phenibut:

Phenibut, originally developed in the Soviet Union in the 1960’s, is a GABA (γ-aminobutyric acid) analogue, which consists of the naturally occurring endogenous amino acid neurotransmitter GABA, with a phenyl group. This allows phenibut to cross the blood brain barrier, whereas GABA supplementation cannot. Phenibut is classified as a central nervous system depressant, but uniquely, it promotes both anxiolytic effects and stimulant effects. Lower concentrations of phenibut are responsible for the stimulant effects, by causing a release of dopamine. In higher concentrations, phenibut acts as more of a sedative. Phenibut, such as phenibut from ELV Bioscience, is in HCL salt form. This is the form that has been most widely studied. There are other forms of phenibut such as free amino acid (Phenibut FAA) and 4-Fluorophenibut (F-Phenibut) but these forms have not been as well studied or used.
How Phenibut Works?
Phenibut has been shown to bind to and activate GABA-b receptors in the brain, similar to the way baclofen works. In very high concentrations, phenibut has also shown to bind to GABA-a receptor, which are the same receptors that benzodiazepines and alcohol (ethanol) bind to. Phenibut research has also shown that it blocks α2δ subunit-containing voltage-gated calcium channels which is why phenibut effects are different from baclofen, for example. Phenibut research has shown that it has a half-life of approximately 5 hours, with an onset time following oral administration of approximately 2-3 hours.
Phenibut Benefits:
Phenibut has been shown to be effective in reducing anxiety and stress, without sedation, at low to moderate doses. In higher doses, phenibut has been shown to relieve insomnia. Other available research and phenibut reviews report that it can be effective for mitigating feelings of depression, and subsequently, for promoting a sense of well-being.
Phenibut Side Effects:
Common phenibut side effects, typically when used in excess, can include agitation, anxiety, headache, hangover, and nausea.
Tianeptine:

Tianeptine, often in the form of tianeptine sodium such as the ELV Bioscience Tianeptine product, is classified as a Selective Serotonin Reuptake Enhancer. However, tianeptine possesses activity at other receptors in the brain which contributes to the overall effects of tianeptine. Research has shown that tianeptine has anti-depressant and anxiolytic properties. Although tianeptine has shown to have anxiolytic effects, it has not shown to have anticholinergic or sedative properties, nor has tianeptine been linked to cardiovascular side effects.
How Tianeptine Works?
As Selective Serotonin Reuptake Enhancer, tianeptine is a positive allosteric modulator of SERT (serotonin transporter), but unlike similar compounds with this action, tianeptine appears to enhance the reuptake of serotonin, rather than inhibit it like tricyclic anti-depressants do. Further tianeptine research to determine its mechanism of action has shown that it is unlikely that tianeptine’s primary mechanism of action is from modulating serotonin levels. Tianeptine has been shown to be a full agonist of the mu-opioid receptor, and to a much lesser extent, a full agonist of the delta-opioid receptor. It is believed that this is the primary mechanism of action whereby tianeptine has shown to possess anti-depressant effects. Unlike traditional opioid receptor agonists, available research has not shown tianeptine to result in tolerance or withdrawal. Tianeptine is also active at the AMPA and NMDA receptors, which blocks some of the effects of glutamate. This effect has shown to create a release in BDNF. Despite tianeptine’s classification as a Selective Serotonin Reuptake Inhibitor, research has shown tianeptine to increase dopamine release. Research has shown tianeptine to be highly bioavailable upon oral administration, with a half-life of approximately 3 hours. Tianeptine is primarily metabolized in the liver.
Tianeptine Benefits:
Although tianeptine has a complex and not fully understood mechanism of action, research and tianeptine reviews have suggested that it has many benefits, including anxiety and stress relief, improvements in mood and sense of well-being, as well as potential pain-relieving and anticonvulsant effects. By increasing BDNF, tianeptine has been shown in animal research to potentially protect against stress-related neurochemical and neuronal changes in the brain.
Tianeptine Side Effects:
Common side effects of tianeptine have been shown to include nausea, headache, dizziness, drowsiness, constipation, and dry mouth.
DMAA (1,3-dimethylamylamine):
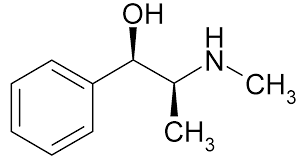
DMAA, also known as methylhexanamine, dimethylamine, and 1,3-DMAA is a sympathetic nervous system stimulant, originally intended as a nasal decongestant. It is available from ELV Bioscience in powder or liquid form. DMAA has been tremendously popular as an energy-boosting supplement, with reported appetite suppressing and mood-enhancing properties. DMAA reviews often suggest that it is used in conjunction with caffeine to enhance its stimulatory and thermogenic effects. DMAA was sold as a dietary supplement in many weight loss and energy products, and some products still contain DMAA, despite the FDA warning against its use in dietary supplements. Many of these supplement companies claim that DMAA could be naturally found in the oil of the geranium plant, but very little, if any, evidence exists to show this to be true.
How DMAA Works?
DMAA stimulates the sympathetic nervous system, much like ephedrine and adrenaline, causing vasoconstriction and bronchodilation. Research has shown that following oral administration, DMAA has a half-life of approximately 8.5 hours.
DMAA Benefits:
Reviews and reports on DMAA use suggest that some find its benefits to include increased physical performance and endurance, increased energy, appetite suppression, thermogenesis and lipolysis (fat loss), as well as improved mood and focus.
DMAA Side Effects:
Reports of DMAA side effects include increased blood pressure, increased heart rate, anxiety, irritability, and a crash following use. DMAA in excessive doses has been shown the potential to create adverse cardiovascular events.
DMHA (Octodrine):

DMHA is often referred to as, 2-Amino-6-methyl heptane, 6-Methyl-2-heptylamine, 2-Aminoisoheptane, and 1, 5-Dimethylhexylamine. DMHA is available from ELV Bioscience in powder and liquid form.
How DMHA Works?
DMHA is a stimulant with a similar chemical structure to DMAA. DMHA has become popular in dietary supplements as a replacement for DMAA. DMHA has a half-life of approximately 9 hours.
DMHA Benefits:
There is little research available on DMHA, but DMHA reviews and reports suggest that it has similar effects to DMAA, but it is less potent on a milligram per milligram basis. These reviews and reports suggest DMHA is often used for performance enhancement, energy, weight loss, and improvements in mood and focus.
DMHA Side Effects:
Due to DMHA’s similarity to DMAA, similar side effects have been reported, such as increased heart rate, blood pressure, and vasoconstriction. Very little research currently exists on DMHA.
Nicotine:
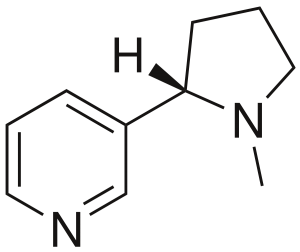
Nicotine is the principal alkaloid in the tobacco plant, which belongs to the nightshade family, which also includes potatoes, tomatoes, and eggplants. Although a subject of much controversy, many people report that nicotine provides nootropic benefits. Of course, the nootropic community’s interest in nicotine is related to routes of administration other than smoking, such as lozenges and gum. Nicotine has been used for centuries as a natural stimulant by way of cultures around the world. Before the appearance of cigarettes and cigars, human beings used to bite on tobacco leaves. While more research is needed, existing research does not draw significant correlations between nicotine itself and cancer. Smoking on the other hand is the primary contributor of carcinogens.
How Nicotine Works?
Some available research, in addition to anecdotal reports, suggests that nicotine can promote working memory improvement, alertness, and improvements in mood and concentration. Nicotine binds to nicotinic acetylcholine receptors in the brain, which promotes the release of certain neurotransmitters, such as dopamine. This effect is often why nicotine is considered to be quite addictive. Nicotine has also shown to increase levels of endogenous opioids in the brain (endorphins). Nicotine has a half-life of approximately two hours and is metabolized in the liver by cytochrome P450 enzymes.
REFERENCES:
Mitchell DC, Knight CA, Hockenberry J, Teplansky R, Hartman TJ (January 2014). “Beverage caffeine intakes in the U.S”. Food and Chemical Toxicology. 63: 136–42. doi:10.1016/j.fct.2013.10.042. PMID 24189158.
Nehlig A, Daval JL, Debry G (1992). “Caffeine and the central nervous system: mechanisms of action, biochemical, metabolic and psychostimulant effects”. Brain Research. Brain Research Reviews. 17 (2): 139–70. doi:10.1016/0165-0173(92)90012-B. PMID 1356551.
“Caffeine”. Pubchem Compound. NCBI. Retrieved 16 October 2014. “
Cano-Marquina A, Tarín JJ, Cano A (May 2013). “The impact of coffee on health”. Maturitas. 75 (1): 7–Qi H, Li S (April 2014). “Dose-response meta-analysis on coffee, tea and caffeine consumption with risk of Parkinson’s disease”. Geriatrics & Gerontology International. 14 (2): 430–9. doi:10.1111/ggi.12123. PMID 23879665.
WHO Model List of Essential Medicines (PDF) (18th ed.). World Health Organization. October 2013 [April 2013]. p. 34 [p. 38 of pdf]. Retrieved 23 December 2014.
Heckman MA, Weil J, De Mejia EG (2010). “Caffeine (1, 3, 7-trimethylxanthine) in Foods: A Comprehensive Review on Consumption, Functionality, Safety, and Regulatory Matters”. Journal of Food Science. 75 (3): R77–R87. doi:10.1111/j.1750-3841.2010.01561.x. PMID 20492310.
Bolton S, Null G (1981). “Caffeine: Psychological Effects, Use and Abuse” (PDF). Orthomolecular Psychiatry. 10 (3): 202–211.
“Caffeine: Summary of Clinical Use”. IUPHAR Guide to Pharmacology. The International Union of Basic and Clinical Pharmacology. Retrieved 13 February 2015.
Snel J, Lorist MM (2011). “Effects of caffeine on sleep and cognition”. Progress in Brain Research. 190: 105–17. doi:10.1016/B978-0-444-53817-8.00006-2. ISBN 978-0-444-53817-8. PMID 21531247.
Dulloo AG, Geissler CA, Horton T, Collins A, Miller DS (January 1989). “Normal caffeine consumption: influence on thermogenesis and daily energy expenditure in lean and postobese human volunteers”. The American Journal of Clinical Nutrition. 49 (1): 44–50. doi:10.1093/ajcn/49.1.44. PMID 2912010.
Acheson KJ, Zahorska-Markiewicz B, Pittet P, Anantharaman K, Jéquier E (May 1980). “Caffeine and coffee: their influence on metabolic rate and substrate utilization in normal weight and obese individuals”. The American Journal of Clinical Nutrition. 33 (5): 989–97. doi:10.1093/ajcn/33.5.989. PMID 7369170.
Southward K, Rutherfurd-Markwick KJ, Ali A (August 2018). “The Effect of Acute Caffeine Ingestion on Endurance Performance: A Systematic Review and Meta-Analysis”. Sports Medicine. 48 (8): 1913–1928. doi:10.1007/s40279-018-0939-8. PMID 29876876.
Grgic J, Trexler ET, Lazinica B, Pedisic Z (2018). “Effects of caffeine intake on muscle strength and power: a systematic review and meta-analysis”. Journal of the International Society of Sports Nutrition. 15: 11. doi:10.1186/s12970-018-0216-0. PMC 5839013. PMID 29527137.
Winston AP (2005). “Neuropsychiatric effects of caffeine”. Advances in Psychiatric Treatment. 11 (6): 432–439. doi:10.1192/apt.11.6.432.
“Caffeine in the diet”. MedlinePlus, US National Library of Medicine. 30 April 2013. Retrieved 2 January 2015.
Smith A (September 2002). “Effects of caffeine on human behavior”. Food and Chemical Toxicology. 40 (9): 1243–55. doi:10.1016/S0278-6915(02)00096-0. PMID 12204388.
Vilarim MM, Rocha Araujo DM, Nardi AE (August 2011). “Caffeine challenge test and panic disorder: a systematic literature review”. Expert Review of Neurotherapeutics. 11 (8): 1185–95. doi:10.1586/ern.11.83. PMID 21797659.
Wang L, Shen X, Wu Y, Zhang D (March 2016). “Coffee and caffeine consumption and depression: A meta-analysis of observational studies”. The Australian and New Zealand Journal of Psychiatry. 50 (3): 228–42. doi:10.1177/0004867415603131. PMID 26339067.
Bruce MS, Lader M (February 1989). “Caffeine abstention in the management of anxiety disorders”. Psychological Medicine. 19 (1): 211–4. doi:10.1017/S003329170001117X. PMID 2727208.
Fredholm BB, Bättig K, Holmén J, Nehlig A, Zvartau EE (March 1999). “Actions of caffeine in the brain with special reference to factors that contribute to its widespread use”. Pharmacological Reviews. 51 (1): 83–133. PMID 10049999
Grosso G, Micek A, Castellano S, Pajak A, Galvano F (January 2016). “Coffee, tea, caffeine and risk of depression: A systematic review and dose-response meta-analysis of observational studies”. Molecular Nutrition & Food Research. 60 (1): 223–34. doi:10.1002/mnfr.201500620. PMID 26518745.
Fredholm BB, Bättig K, Holmén J, Nehlig A, Zvartau EE (March 1999). “Actions of caffeine in the brain with special reference to factors that contribute to its widespread use”. Pharmacological Reviews. 51 (1): 83–133. PMID 10049999.
Santos C, Costa J, Santos J, Vaz-Carneiro A, Lunet N (2010). “Caffeine intake and dementia: systematic review and meta-analysis”. Journal of Alzheimer’s Disease. 20 Suppl 1: S187–204. doi:10.3233/JAD-2010-091387. PMID 20182026.
Panza F, Solfrizzi V, Barulli MR, Bonfiglio C, Guerra V, Osella A, Seripa D, Sabbà C, Pilotto A, Logroscino G (March 2015). “Coffee, tea, and caffeine consumption and prevention of late-life cognitive decline and dementia: a systematic review”. The Journal of Nutrition, Health & Aging. 19 (3): 313–28. doi:10.1007/s12603-014-0563-8. PMID 25732217.
Winston AP, Hardwick E, Jaberi N (2005). “Neuropsychiatric effects of caffeine”. Advances in Psychiatric Treatment. 11 (6): 432–439. doi:10.1192/apt.11.6.432. Retrieved 19 December 2013.
Benowitz NL (1990). “Clinical pharmacology of caffeine”. Annual Review of Medicine. 41: 277–88. doi:10.1146/annurev.me.41.020190.001425. PMID 2184730.
“Caffeine (Systemic)”. MedlinePlus. 25 May 2000. Archived from the original on 23 February 2007. Retrieved 3 August 2009.
Froestl W, Muhs A, Pfeifer A (2012). “Cognitive enhancers (nootropics). Part 1: drugs interacting with receptors”. Journal of Alzheimer’s Disease. 32 (4): 793–887. doi:10.3233/JAD-2012-121186. PMID 22886028.
“Caffeine”. IUPHAR. International Union of Basic and Clinical Pharmacology. Retrieved 2 November 2014.
Fisone G, Borgkvist A, Usiello A (April 2004). “Caffeine as a psychomotor stimulant: mechanism of action”. Cellular and Molecular Life Sciences. 61 (7–8): 857–72. doi:10.1007/s00018-003-3269-3. PMID 15095008.
Ferré S, Bonaventura J, Tomasi D, Navarro G, Moreno E, Cortés A, Lluís C, Casadó V, Volkow ND (May 2016). “Allosteric mechanisms within the adenosine A2A-dopamine D2 receptor heterotetramer”. Neuropharmacology. 104: 154–60. doi:10.1016/j.neuropharm.2015.05.028. PMC 5754196. PMID 26051403.
An update on the mechanisms of the psychostimulant effects of caffeine. J. Neurochem.. 2008;105(4):1067–1079. doi:10.1111/j.1471-4159.2007.05196.x. PMID 18088379. “On the other hand, our ‘ventral shell of the nucleus accumbens’ very much overlaps with the striatal compartment…”
Ribeiro JA, Sebastião AM (2010). “Caffeine and adenosine”. Journal of Alzheimer’s Disease. 20 Suppl 1: S3–15. doi:10.3233/JAD-2010-1379. PMID 20164566.Teekachunhatean S, Tosri N, Rojanasthien N, Srichairatanakool S, Sangdee C (8 January 2013). “Pharmacokinetics of Caffeine following a Single Administration of Coffee Enema versus Oral Coffee Consumption in Healthy Male Subjects”. ISRN Pharmacology. 2013 (147238): 147238. doi:10.1155/2013/147238. PMC 3603218. PMID 23533801.
Williams R (21 September 2016). “How Plants Evolved Different Ways to Make Caffeine”.
“Caffeine Content of Food and Drugs”. Nutrition Action Health Newsletter. Center for Science in the Public Interest. 1996. Archived from the original on 14 June 2007. Retrieved 3 August 2009.
Smit HJ, Gaffan EA, Rogers PJ (November 2004). “Methylxanthines are the psycho-pharmacologically active constituents of chocolate”. Psychopharmacology. 176 (3–4): 412–9. doi:10.1007/s00213-004-1898-3. PMID 15549276.
Bempong DK, Houghton PJ, Steadman K (1993). “The xanthine content of guarana and its preparations”. Int J Pharmacog. 31 (3): 175–181. doi:10.3109/13880209309082937. ISSN 0925-1618.
Rätsch, Christian (2005). The Encyclopedia of Psychoactive Plants: Ethnopharmacology and Its Applications. Simon and Schuster. p. PT1235. ISBN 9781594776625.
“Theanine”. Pubchem Compound. NCBI. Retrieved 21 February 2015.
Weinberg BA, Bealer BK (2001). The World of caffeine: The Science and Culture of the World’s Most Popular Drug. Routledge. p. 195. ISBN 978-0-415-92723-9. Retrieved 15 January 2014.
Nathan, Pradeep; Lu, Kristy; Gray, M.; Oliver, C. (2006). “The Neuropharmacology of L-Theanine(N-Ethyl-L-Glutamine)”. Journal of Herbal Pharmacotherapy. 6 (2): 21–30. doi:10.1300/J157v06n02_02. PMID 17182482.
“D-theanine | C7H14N2O3”. ChemSpider.com. Retrieved 2015-05-21.
Kakuda T (2002). “Neuroprotective effects of the green tea components theanine and catechins”. Biol. Pharm. Bull. 25 (12): 1513–8. doi:10.1248/bpb.25.1513. PMID 12499631.
Narukawa M, Toda Y, Nakagita T, Hayashi Y, Misaka T (2014). “L-Theanine elicits umami taste via the T1R1 + T1R3 umami taste receptor”. Amino Acids. 46 (6): 1583–7. doi:10.1007/s00726-014-1713-3. PMID 24633359.
Kakuda T (2011). “Neuroprotective effects of theanine and its preventive effects on cognitive dysfunction”. Pharmacol. Res. 64 (2): 162–8. doi:10.1016/j.phrs.2011.03.010. PMID 21477654.
Wakabayashi C, Numakawa T, Ninomiya M, Chiba S, Kunugi H (2012). “Behavioral and molecular evidence for psychotropic effects in L-theanine”. Psychopharmacology. 219 (4): 1099–109. doi:10.1007/s00213-011-2440-z. PMID 21861094.
Yokogoshi H, Kobayashi M, Mochizuki M, Terashima T (1998). “Effect of theanine, r-glutamylethylamide, on brain monoamines and striatal dopamine release in conscious rats”. Neurochem. Res. 23 (5): 667–73. doi:10.1023/A:1022490806093. PMID 9566605.
Kimura, Kenta; Ozeki, Makoto; Juneja, Lekh Raj; Ohira, Hideki (2007). “L-Theanine reduces psychological and physiological stress responses”. Biological Psychology. 74 (1): 39–45. doi:10.1016/j.biopsycho.2006.06.006. PMID 16930802.
Yokogoshi, Hidehiko; Mochizuki, Mikiko; Saitoh, Kotomi (1998). “Theanine-induced Reduction of Brain Serotonin Concentration in Rats”. Bioscience, Biotechnology, and Biochemistry. 62 (4): 816–7. doi:10.1271/bbb.62.816. PMID 9614715.
Owen, Gail N.; Parnell, Holly; De Bruin, Eveline A.; Rycroft, Jane A. (2008). “The combined effects of L-theanine and caffeine on cognitive performance and mood”. Nutritional Neuroscience. 11 (4): 193–8. doi:10.1179/147683008X301513. PMID 18681988.
Haskell, Crystal F.; Kennedy, David O.; Milne, Anthea L.; Wesnes, Keith A.; Scholey, Andrew B. (2008). “The effects of l-theanine, caffeine and their combination on cognition and mood”. Biological Psychology. 77 (2): 113–22. doi:10.1016/j.biopsycho.2007.09.008. PMID 18006208.
Park, Sang-Ki; Jung, In-Chul; Lee, Won Kyung; Lee, Young Sun; Park, Hyoung Kook; Go, Hyo Jin; Kim, Kiseong; Lim, Nam Kyoo; et al. (2011). “A Combination of Green Tea Extract andl-Theanine Improves Memory and Attention in Subjects with Mild Cognitive Impairment: A Double-Blind Placebo-Controlled Study”. Journal of Medicinal Food. 14 (4): 334–43. doi:10.1089/jmf.2009.1374. PMID 21303262.
Bryan, Janet (2008). “Psychological effects of dietary components of tea: Caffeine and L-theanine”. Nutrition Reviews. 66 (2): 82–90. doi:10.1111/j.1753-4887.2007.00011.x. PMID 18254874.
Raloff, Janet (September 29, 2007). “Distracted? Tea might help your focus”. Science News. 172 (13): 206. doi:10.1002/scin.2007.5591721319.
Giesbrecht, T.; Rycroft, J.A.; Rowson, M.J.; De Bruin, E.A. (2010). “The combination of L-theanine and caffeine improves cognitive performance and increases subjective alertness”. Nutritional Neuroscience. 13 (6): 283–90. doi:10.1179/147683010X12611460764840. PMID 21040626.
Kelly, Simon P.; Gomez-Ramirez, Manuel; Montesi, Jennifer L.; Foxe, John J. (2008). “L-Theanine and Caffeine in Combination Affect Human Cognition as Evidenced by Oscillatory alpha-Band Activity and Attention Task Performance”. The Journal of Nutrition. 138 (8): 1572S–1577S. PMID 18641209.
Nathan, PJ; Lu, K; Gray, M; Oliver, C (2015-04-20). “The neuropharmacology of L-theanine(N-ethyl-L-glutamine): a possible neuroprotective and cognitive enhancing agent”. J Herb Pharmacother. 6 (2): 21–30. doi:10.1300/J157v06n02_02. PMID 17182482.
Keenan, Emma K.; Finnie, Mike D.A.; Jones, Paul S.; Rogers, Peter J.; Priestley, Caroline M. (2011). “How much theanine in a cup of tea? Effects of tea type and method of preparation”. Food Chemistry. 125 (2): 588. doi:10.1016/j.foodchem.2010.08.071.
Zvejniece L, Vavers E, Svalbe B, Veinberg G, Rizhanova K, Liepins V, Kalvinsh I, Dambrova M (2015). “R-phenibut binds to the α2-δ subunit of voltage-dependent calcium channels and exerts gabapentin-like anti-nociceptive effects”. Pharmacol. Biochem. Behav. 137: 23–9. doi:10.1016/j.pbb.2015.07.014. PMID 26234470.
Lapin, Izyaslav (7 June 2006). “Phenibut (β-Phenyl-GABA): A Tranquilizer and Nootropic Drug”. CNS Drug Reviews. 7 (4): 471–481. doi:10.1111/j.1527-3458.2001.tb00211.x. ISSN 1080-563X.
Froestl W (2010). Chemistry and pharmacology of GABAB receptor ligands. Adv. Pharmacol. Advances in Pharmacology. 58. pp. 19–62. doi:10.1016/S1054-3589(10)58002-5. ISBN 9780123786470. PMID 20655477.
Lapin, I. (2001). “Phenibut (beta-phenyl-GABA): A tranquilizer and nootropic drug”. CNS Drug Reviews. 7 (4): 471–481. doi:10.1111/j.1527-3458.2001.tb00211.x. PMID 11830761.
Vavers E, Zvejniece L, Svalbe B, Volska K, Makarova E, Liepinsh E, Rizhanova K, Liepins V, Dambrova M (2016). “The neuroprotective effects of R-phenibut after focal cerebral ischemia”. Pharmacol. Res. 113 (Pt B): 796–801. doi:10.1016/j.phrs.2015.11.013. PMID 26621244.
Zyablitseva, Evgeniya A.; Kositsyn, Nikolay S.; Shul’gina, Galina I. (2013). “The Effects of Agonists of Ionotropic GABAA and Metabotropic GABAB Receptors on Learning”. The Spanish Journal of Psychology. 12 (1): 12–20. doi:10.1017/S1138741600001438. ISSN 1138-7416. PMID 19476215.
Wagstaff, AJ; Ormrod, D; Spencer, CM (March 2001). “Tianeptine A Review of its Use in Depressive Disorders”. CNS Drugs. 15 (3): 231–259. doi:10.2165/00023210-200115030-00006. PMID 11463130.
McEwen, BS; Chattarji, S; Diamond, DM; Jay, TM; Reagan, LP; Svenningsson, P; Fuchs, E (March 2010). “The neurobiological properties of tianeptine (Stablon): from monoamine hypothesis to glutamatergic modulation”. Molecular Psychiatry. 15 (3): 237–49. doi:10.1038/mp.2009.80. PMC 2902200. PMID 19704408.
McEwen, BS; Olié, JP (June 2005). “Neurobiology of mood, anxiety, and emotions as revealed by studies of a unique antidepressant: tianeptine”. Molecular Psychiatry. 10 (6): 525–37. doi:10.1038/sj.mp.4001648. PMID 15753957.
McEwen, BS; Chattarji, S (December 2004). “Molecular mechanisms of neuroplasticity and pharmacological implications: the example of tianeptine”. European Neuropsychopharmacology. 14 Suppl 5: S497–502. doi:10.1016/j.euroneuro.2004.09.008. PMID 15550348.
Kasper, S; McEwen, BS (2008). “Neurobiological and clinical effects of the antidepressant tianeptine”. CNS Drugs. 22 (1): 15–26. doi:10.2165/00023210-200822010-00002. PMID 18072812.
Brink, CB; Harvey, BH; Brand, L (January 2006). “Tianeptine: a novel atypical antidepressant that may provide new insights into the biomolecular basis of depression”. Recent Patents on CNS Drug Discovery. 1 (1): 29–41. doi:10.2174/157488906775245327. PMID 18221189. Archived from the original on 14 April 2013.
Gassaway MM, Rives ML, Kruegel AC, Javitch JA, Sames D (2014). “The atypical antidepressant and neurorestorative agent tianeptine is a μ-opioid receptor agonist”. Transl Psychiatry. 4: e411. doi:10.1038/tp.2014.30. PMC 4119213. PMID 25026323.
Defrance, R; Marey, C; Kamoun, A (1988). “Antidepressant and anxiolytic activities of tianeptine: an overview of clinical trials” (PDF). Clinical Neuropharmacology. 11 Suppl 2: S74–82. PMID 2902922. Archived from the original (PDF) on 4 April 2016.
Baune, BT; Renger, L (September 2014). “Pharmacological and non-pharmacological interventions to improve cognitive dysfunction and functional ability in clinical depression – A systematic review”. Psychiatry Research. 219 (1): 25–50. doi:10.1016/j.psychres.2014.05.013. PMID 24863864.
Mennini, T; Mocaer, E; Garattini, S (1987). “Tianeptine, a selective enhancer of serotonin uptake in rat brain”. Naunyn Schmiedebergs Arch Pharmacol. 336 (5): 478–482. doi:10.1007/bf00169302. PMID 3437921.
Tianeptine Sodium. Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. 5 December 2011. Retrieved 2 December 2013.
Waintraub, L; Septien L; Azoulay, P (January 2002). “Efficacy and safety of tianeptine in major depression: evidence from a 3-month controlled clinical trial versus paroxetine”. CNS Drugs. 16 (1): 65–75. doi:10.2165/00023210-200216010-00005. PMID 11772119.
Uzbay, TI (May 2008). “Tianeptine: potential influences on neuroplasticity and novel pharmacological effects”. Progress in Neuro-psychopharmacology & Biological Psychiatry. 32 (4): 915–24. doi:10.1016/j.pnpbp.2007.08.007. PMID 17826881.
Bailey, Sarah J.; Almatroudi, Abdulrahman; Kouris, Andreas (2018). “Tianeptine: An Atypical Antidepressant with Multimodal Pharmacology”. Current Psychopharmacology. 6 (2). doi:10.2174/2211556006666170525154616. ISSN 2211-5560.
Alici, T; Kayir, H; Aygoren, MO; Saglam, E; Uzbay, IT (January 2006). “Discriminative stimulus properties of tianeptine”. Psychopharmacology. 183 (4): 446–51. doi:10.1007/s00213-005-0210-5. PMID 16292591.
Cavalla, D; Chianelli, F (August 2015). “Tianeptine prevents respiratory depression without affecting analgesic effect of opiates in conscious rats”. European Journal of Pharmacology. doi:10.1016/j.ejphar.2015.05.067. PMID 26068549.
Samuels BA, Nautiyal KM, Kruegel AC, Levinstein MR, Magalong VM, Gassaway MM, Grinnell SG, Han J, Ansonoff MA, Pintar JE, Javitch JA, Sames D, Hen R (2017). “The Behavioral Effects of the Antidepressant Tianeptine Require the Mu Opioid Receptor”. Neuropsychopharmacology. doi:10.1038/npp.2017.60. PMID 28303899.
Invernizzi, R; Pozzi, L; Garattini, S; Samanin, R (March 1992). “Tianeptine increases the extracellular concentrations of dopamine in the nucleus accumbens by a serotonin-independent mechanism”. Neuropharmacology. 31 (3): 221–7. doi:10.1016/0028-3908(92)90171-K. PMID 1630590.
Venhuis, Bastiaan J.; De Kaste, Dries (2012). “Scientific opinion on the regulatory status of 1,3-Dimethylamylamine (DMAA)”. European Journal of Food Research & Review. 2 (4): 93–100.
“1,3-Dimethylpentylamine – Compound Summary”. PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Identification and Related Records. Retrieved 27 May 2012.
Cohen, Pieter A. (8 April 2013). “DMAA as a Dietary Ingredient—Reply”. JAMA Internal Medicine. 173 (7): 594. doi:10.1001/jamainternmed.2013.3776.
Lisi A, Hasick N, Kazlauskas R, Goebel C (2011). “Studies of methylhexaneamine in supplements and geranium oil”. Drug Test Anal. 3 (11–12): 873–6. doi:10.1002/dta.392. PMID 22147493.
Heather L. Fleming, Patricia L. Ranaivo and Paul S. Simone (2012). “Analysis and Confirmation of 1,3-DMAA and 1,4-DMAA in Geranium Plants Using High Performance Liquid Chromatography with Tandem Mass Spectrometry at ng/g Concentrations”. Analytical Chemistry Insights (7): 59–78. doi:10.4137/ACI.S10445. PMC 3512447.
Second Edition, Reprint Edition, Volume 1-2. 1988 Noyes Publications. Westwood, New Jersey.
Miya, TS; Edewards, LD (Feb 1953). “A pharmacological study of certain alkoxyalkylamines”. J Am Pharm Assoc Am Pharm Assoc (Baltim). 42 (2): 107–10. PMID 13034643.
“Methylhexaneamine Carbonate” entry in Marshall Sittig. Pharmaceutical Manufacturing Encyclopedia,
Cohen, Pieter A.; Travis, John C.; Keizers, Peter H. J.; Deuster, Patricia; Venhuis, Bastiaan J. (8 November 2017). “Four experimental stimulants found in sports and weight loss supplements: 2-amino-6-methylheptane (octodrine), 1,4-dimethylamylamine (1,4-DMAA), 1,3-dimethylamylamine (1,3-DMAA) and 1,3-dimethylbutylamine (1,3-DMBA)”. Clinical Toxicology. 56 (6): 421–426. doi:10.1080/15563650.2017.1398328.
“Nicotinic acetylcholine receptors: Introduction”. IUPHAR Database. International Union of Basic and Clinical Pharmacology. Retrieved 1 September 2014.
CID 10982 from PubChem
Mineur YS, Picciotto MR (December 2010). “Nicotine receptors and depression: revisiting and revising the cholinergic hypothesis”. Trends in Pharmacological Sciences. 31 (12): 580–6. doi:10.1016/j.tips.2010.09.004. PMC 2991594. PMID 20965579.
“Does nicotine cause cancer?”. European Code Against Cancer. World Health Organization – International Agency for Research on Cancer. Retrieved 23 January 2019.
Kishioka S, Kiguchi N, Kobayashi Y, Saika F (2014). “Nicotine effects and the endogenous opioid system”. Journal of Pharmacological Sciences. 125 (2): 117–24. doi:10.1254/jphs.14R03CP. PMID 24882143.
Henningfield JE, Calvento E, Pogun S (2009). Nicotine Psychopharmacology. Springer. pp. 35, 37. ISBN 978-3-540-69248-5.
Sarter M (August 2015). “Behavioral-Cognitive Targets for Cholinergic Enhancement”. Current Opinion in Behavioral Sciences. 4: 22–26. doi:10.1016/j.cobeha.2015.01.004. PMC 5466806. PMID 28607947.
